Construction of a Winogradsky Column
Student Version (go to Instructor Version)
| Subject Area(s) | microbiology |
| Intended Audience |
high school biology, independent study/science fair, introductory undergraduate microbiology, advanced college level microbiology. |
| Type | Laboratory Exercise |
| Revision Date | June 18, 2004 |
CONTENT
Sergei Winogradsky (1856-1953) was among the first microbiologists to investigate the organisms found in complex biofilm communities, providing some of the first descriptions of sulfur, iron and nitrogen metabolizing bacteria as well as those bacteria involved in cellulose decomposition.Working in the late decades of the last century, Winogradsky was a pioneer in selective enrichment techniques. One of the strategies that he employed to isolate organisms from nature was a miniature model pond cross section which has since been called a Winogradsky column.

MATERIALS AND EQUIPMENT
Quantity |
Description |
| As Necessary | a source of soil or mud |
| As Necessary | a source of water (same source as the mud or other, tap water although it contains a residue of chlorine has been used successfully) . |
| As Necessary | rubber or vinyl gloves |
| As Necessary | metal or plastic pans for mixing mud with other ingredients |
| As Necessary | spatulas |
| As Necessary | sources of carbon (i.e. some, such as calcium carbonate and chalk are rapidly metabolized while others such as shredded paper and saw dust are metabolized more slowly) |
| As Necessary | sources of sulfur (e.g. elemental sulfur, sodium sulfate, raw or hard boiled eggs, sodium sulfide) |
| As Necessary | a bottle (2 liter soda bottle) or other tall, clear container (wide mouth containers are preferable). |
DETAILED PROCEDURE
- Mix the mud/soil with the desired carbon and sulfur sources.
- Add some water to the container and add the mud/soil mixture a little at a time using a glass rod or stick to pack the material in the bottom of the column. Air bubbles should be excluded as much as possible, although any oxygen remaining trapped in the mud is probably consumed by facultative organisms very rapidly.
- Add water and soil mixture until the column is about three fourths full. Use a wash bottle to rinse excess mud from the container above the soil level and then add additional water to produce a water column (the pond). Leave some air space above the surface of the water and cover the column with plastic wrap or parafilm. Be sure to cut a slit or two in the plastic wrap or parafilm to permit gas exchange.
- Place the column near a window or tungsten lamp in a well ventilated room. They can smell until they stabilize. Check to make sure that the column is not heated too much by the light. The temperature of the column should not rise much above room temperature.
- Over the next several weeks, observe any changes in the column (color, gas bubbles, odor). Record these observations in your notebook. Note: Polaroid photographs taken at regular intervals are a great way to record changes in color and color distribution with in the column.
Possible carbon sources:
Vegetable materials such as shredded hay, newsletter or sawdust, grass clippings, corn starch, and oatmeal will work fine. Use your imagination. These materials release carbon relatively slowly. Fast release carbon sources include sodium carbonate, sodium bicarbonate and calcium carbonate.
Possible sulfur sources:
Once again use your imagination. Elemental sulfur, calcium sulfate, magnesium sulfate, raw or hard-boiled eggs, and cheese have all been used with success.
SAFETY NOTE: There is no easy way to determine if the mud is contaminated with sewage, farm waste, heavy metals or other toxic materials. Wear latex or vinyl gloves when mixing materials and packing the column.
Column structure and physiology:
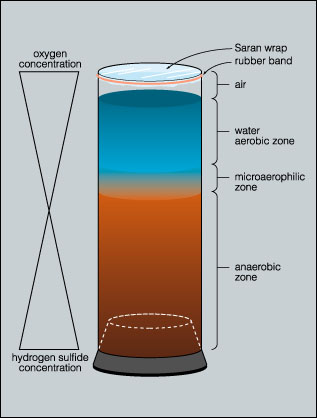
The water column at the surface is in contact with the atmosphere and is therefore aerobic but it becomes increasingly anaerobic with depth. The surface layer of the column may produce an aerobic liquid air biofilm (pellicle) that can be sampled by dipping a coverslip into the column and lifting a portion of the film from the water.
In the highly anaerobic base of the column, decomposition and the activity of sulfate reducing bacteria results in the production of hydrogen sulfide gas. This hydrogen sulfide gradient decreases toward the top of the column. These two gradients (oxygen and hydrogen sulfide), acting in opposite directions, create a continuous range of habitats selective for a variety of microorganisms.
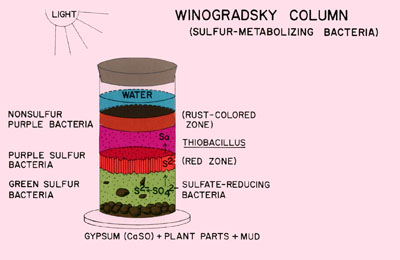
Typically the lower portions of the column are colonized by photoautotrophic green and purple sulfur bacteria. The aerobic surface of the column is occupied by oxygenic cyanobacteria. Just below the surface phototrophic purple non-sulfur bacteria predominate.
The majority of the bacteria are located in a thin film between the soil/mud substrate and the container wall. When using plastic containers such as two liter pop bottles, these bacteria can be sampled by inserting a needle through the container wall or by freezing the column and cutting into the column with a utility knife and shears to expose the surface layer of the column.
ASSESSMENT / EVALUATION
The success of the column will be evident within a few weeks with the appearance of dramatically colored biofilms of phototrophic green and purple sulfur bacteria just under the glass or plastic layer of the column. At the top of the water column, green algae and cyanobacteria will appear.
FOLLOW-UP ACTIVITIES AND QUESTIONS
- Light effects - Have some students occlude certain regions of the bottle so that light cannot penetrate (use aluminum foil, or cardboard masking). Does this affect the appearance of the column?
- More light effects - Have students explore the absorption spectra of the various types of phototrophs likely to grow at each region of the (consult with your theater department for colored gels used in lighting).
- Recovery of phototrophic bacteria - Carolina Biological sells Winogradsky column kits (FA-70-3490). These use a cylindrical plastic column with a rubber stopper base. These kits are quite convenient and easy to set up. It is possible to recover phototrophic bacteria from a column by freezing the established column and then pushing out the frozen plug into a pan. Microorganisms for microscopic observation or further culturing may be obtained from the plug surface. This could also be done by cutting into a frozen column in a plastic 2 L soda bottle. With care a utility knife or pair of heavy shears could be used to expose the column surface.
- Electrochemical potential – Cut two lengths of bell cord wire, one long enough to reach the bottom of the column and the other just long enough to touch the surface of the mud below the water layer. Strip the last cm of insulation from the ends of each wire. Place the long wire in the column so that the lower end rests at the bottom of the column. Place the short wire so that it reaches the surface of the mud below the water column. Bend both wires so that they hang over the top edge of the column. Connect the wires to a VOM (Volt Ohm Meter) set to measure voltage in mVolts. What is the source of the electrical current? Is it chemical or photochemical? How could you decide? Could the world energy crisis be cured by drawing energy from mud flats and bogs?
REFERENCES:
Additional instructions for constructing Winogradsky columns can be found in many laboratory manuals, and in informative Carolina Tips (Sept. 1, 1978) and on the Internet.
This material is based upon work supported by the National Science Foundation under Grant No. 0618744, and in part by the Waksman Foundation for Microbiology. Developed in collaboration with Dr. John Lennox, Penn State Altoona. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation.
©2002-2008 Center for Biofilm Engineering, http://www.biofilm.montana.edu
