Biofilm Flow Reactor
A substantially updated version of the hypertextbook is available here. Please migrate to that version. This one will eventually disappear.
Copyright © Alfred B. Cunningham, John E. Lennox, and Rockford J. Ross, Eds. 2001-2008
Building and Using a Biofilm Continuous Flow Stirred Reactor
Supplies Needed
Quantity |
Description |
| 1 | wide mouth polycarbonate sample jar with cap |
| 3 | #1 (1-holed) rubber stoppers |
| 4 | #6 rubber stoppers |
| 4 | 1x3 inch microscope slides (1mm thick) |
| 1 | fine tooth modeling saw |
| 1 | magnetic stir bar (a 1 inch teflon bar works well) |
| 360 ml (approx) | 1/10 Nutrient Broth, liquid Luria-Bertani (LB) broth, or any other desired medium |
| 1 | Millipore sterile air vent (50 mm) (Millex-FG 50) |
| 1 | influent port (#1 1-holed rubber stopper with glass tubing inserted for in flow) |
| 1 | effluent port (#1 1-holed rubber stopper with glass tubing inserted for out flow) |
*The ports in the reactor lid can be cut with a laser engraver, drill, or drill press with a 1 1/16 inch paddle-type bit for the larger holes in the lid, and a 1/2 inch paddle-type bit for the smaller holes in the lid and side of the reactor.
Instructions
- Microscope slides are inserted into the slots which have been cut into each of the four # 6 rubber stoppers.
- The slide rubber stopper assemblies are placed into the 1 1/16 inch holes in the lid of the polyethylene jar. Orient the slides in the desired direction. Orientation parallel to the wall of the jar gives a less turbulent flow when the reactor is in operation.
- Insert a glass delivery tube into a #1 (1-holed) rubber stopper. Place this in the center 1/2 inch port of the lid. Using a piece of flexible tubing, attach the sterile Millipore air vent to this module.
- Insert the influent (in flow) glass tube into a #1 (1-holed) rubber stopper. Insert this module into the outermost 1/2 inch port of the lid.
- Insert the effluent (out flow) glass tube into a #1 (1-holed) rubber stopper. Insert this module into the 1/2 inch port on the side of the reactor.
- Sufficient liquid medium of the desired type is added to the jar to cover the slides to the desired depth (about 360 ml).
- A magnetic stir bar is placed in the jar, and the top of the jar is screwed into place.
- The entire apparatus is now sterilized by autoclaving.
- In operation the medium in the reactor is stirred by placing the reactor on a magnetic stirrer. The degree of agitation is controlled by the rotation speed of the magnetic stir bar.
- The reactor may be inoculated through one of the rubber stopper ports.
Companion exercises
- Gram staining.
- Harvesting biofilm associated cells from the glass slides by scraping and sonication and their enumeration by the drop plate method.
Illustration
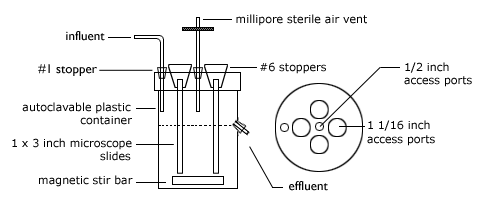
Continuous Flow Stirred Reactor
Educational Program Curricula and Teaching Resources
Supported in part by the Waksman Foundation for MicrobiologyDeveloped in collaboration with Dr. John Lennox, Penn State University-Altoona
©1999-2006 Center for Biofilm Engineering, http://www.erc.montana.edu